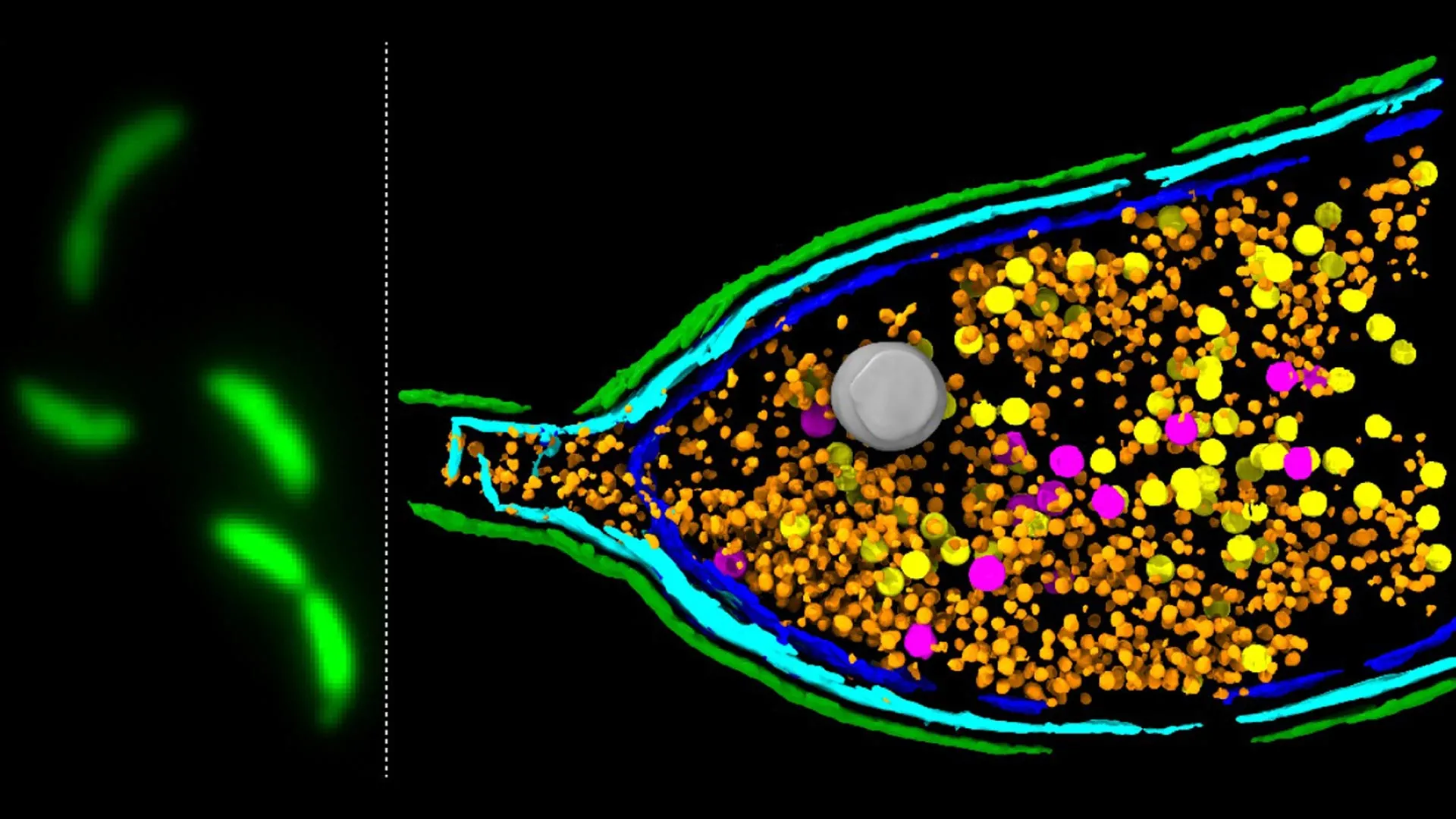
Scientists at the John Innes Centre have made a significant breakthrough in understanding the intricate mechanisms by which bacteria share genetic material, including those responsible for antimicrobial resistance (AMR), a rapidly escalating global health crisis. Their latest research, published in the esteemed journal Nature Microbiology, delves into the operation of unusual viral-like particles known as gene transfer agents (GTAs). These particles, once thought to be mere remnants of ancient viruses, are now revealed to be sophisticated tools actively employed by bacteria for genetic dissemination.
The Sophisticated World of Gene Transfer Agents
Gene transfer agents (GTAs) bear a striking resemblance to bacteriophages, the viruses that specifically infect and replicate within bacterial cells. However, GTAs are not the destructive invaders commonly associated with phages. Instead, they represent a fascinating evolutionary adaptation where bacteria have harnessed components of ancient viral machinery, bringing them under their own genetic command and repurposing them for beneficial purposes. Unlike typical bacteriophages, GTAs do not carry the genetic instructions to replicate themselves within the host cell; rather, they are packaged with fragments of the host bacterium’s own DNA.
These remarkably evolved particles function as highly efficient molecular couriers. They meticulously package random segments of DNA from their host bacterium and then are released to deliver these genetic payloads to neighboring bacterial cells. This fundamental process, known as horizontal gene transfer (HGT), is a cornerstone of bacterial evolution, enabling populations to rapidly acquire and share advantageous traits. Crucially, this includes genes that confer resistance to antibiotics, a phenomenon that is undermining the effectiveness of essential medicines and posing a dire threat to public health worldwide. The World Health Organization (WHO) has designated AMR as one of the top 10 global public health threats facing humanity. It is estimated that by 2050, AMR could cause 10 million deaths annually, surpassing cancer deaths.
Unlocking the Mystery of GTA Release: Host Cell Lysis
A critical and previously poorly understood step in the GTA lifecycle is host cell lysis. This is the process by which the bacterial cell membrane ruptures, releasing the newly assembled GTA particles, laden with DNA, into the surrounding environment. Without this controlled breakdown of the host cell, the GTAs would remain trapped, rendering their gene-sharing function ineffective. Deciphering the precise molecular machinery responsible for this lysis has been a long-standing challenge for researchers.
The team at the John Innes Centre, in collaboration with researchers from the University of York and the Rowland Institute at Harvard, employed a cutting-edge deep sequencing-based screening method. This advanced technique allowed them to systematically identify and analyze genes critical for GTA activity within the well-studied model bacterium Caulobacter crescentus. This meticulous approach enabled them to pinpoint a specific cluster of genes that orchestrates the release of GTAs.
The LypABC System: A Central Control Hub for Lysis
The investigation revealed a pivotal three-gene system, designated LypABC, responsible for producing bacterial proteins that drive cell lysis. Through careful genetic manipulation, the researchers demonstrated that when the lypabc genes were deleted from the bacterial genome, the cells were unable to break open and release GTA particles. Conversely, when the LypABC system was deliberately overactivated, a significantly higher proportion of bacterial cells underwent lysis, facilitating the release of GTAs. These compelling results firmly establish LypABC as a central control hub, meticulously regulating the process of GTA-mediated cell lysis.
A Surprising Repurposing: From Immune Defense to Gene Transfer
Perhaps the most astonishing revelation from this research is the striking similarity between the LypABC system and a known bacterial anti-phage immune mechanism. Bacterial immune systems are designed to detect and neutralize invading viruses, and the protein components within LypABC are typically associated with such defensive roles. However, in this context, the bacteria appear to have ingeniously repurposed this ancient defense system. Instead of solely fighting off viral invaders, the LypABC system has been co-opted to facilitate the release of GTAs, thereby promoting the spread of genetic material among bacterial populations. This remarkable example underscores the extraordinary flexibility and adaptability of bacterial biological systems, demonstrating how existing cellular machinery can be ingeniously redeployed for entirely new functions.
The Imperative of Tight Regulation for Bacterial Survival
The study also identified a crucial regulatory protein that plays a vital role in maintaining strict control over GTA activity. This tight regulation is not merely a matter of efficiency; it is essential for bacterial survival. The researchers found that improper or uncontrolled activation of the LypABC system can be highly toxic to bacterial cells, leading to widespread cell death. Therefore, the precise molecular mechanisms that govern the activation and deactivation of LypABC are critical for the bacterium’s ability to engage in gene transfer without compromising its own integrity. This finding highlights a delicate balancing act that bacteria perform, leveraging powerful genetic exchange mechanisms while ensuring their own continued existence.
Broader Implications for Combating Antimicrobial Resistance
The insights gleaned from this research offer a profound deepening of our understanding of how genetic information moves between bacterial cells. This dynamic process is a primary driver behind the alarming spread of antibiotic resistance. By elucidating the specific molecular pathways involved in GTA-mediated gene transfer, scientists are better equipped to develop novel strategies to disrupt this critical process. Understanding how bacteria "package and deliver" resistance genes could pave the way for new therapeutic interventions aimed at preventing or reversing the spread of AMR.
Dr. Emma Banks, the first author of the study and a Royal Commission for the Exhibition of 1851 Research Fellow, articulated the significance of the findings: "What’s particularly interesting is that LypABC looks like an immune system, yet bacteria are using it to release GTA particles. It suggests that immune systems can be repurposed to help bacteria share DNA with each other — a process that can contribute to the spread of antibiotic resistance."
Future Directions: Unraveling the Activation Triggers
The immediate next step for the research team is to delve deeper into the intricate mechanisms that govern the activation of the LypABC system. Understanding precisely what signals trigger this control hub and how it orchestrates the precise rupture of bacterial cells to release GTA particles will be crucial for fully harnessing this knowledge. This could involve investigating environmental cues, intercellular communication, or specific genetic triggers that initiate the lysis process.
This groundbreaking work not only sheds new light on the sophisticated adaptations of bacteria but also provides invaluable clues in the ongoing global battle against antimicrobial resistance. By understanding the fundamental processes that allow bacteria to exchange genetic material, researchers are moving closer to developing innovative solutions to this pressing public health challenge. The study, titled "A bacterial CARD-NLR-like immune system controls the release of gene transfer agents," represents a significant leap forward in our comprehension of microbial genetics and its profound implications for human health. The implications of this research are far-reaching, potentially informing the development of new antimicrobial strategies that target the very pathways bacteria use to evolve and spread resistance.